Court restricts abortion access across the US by blocking the mailing of mifepristone
✨ AI Summary
🔊 جاري الاستماع
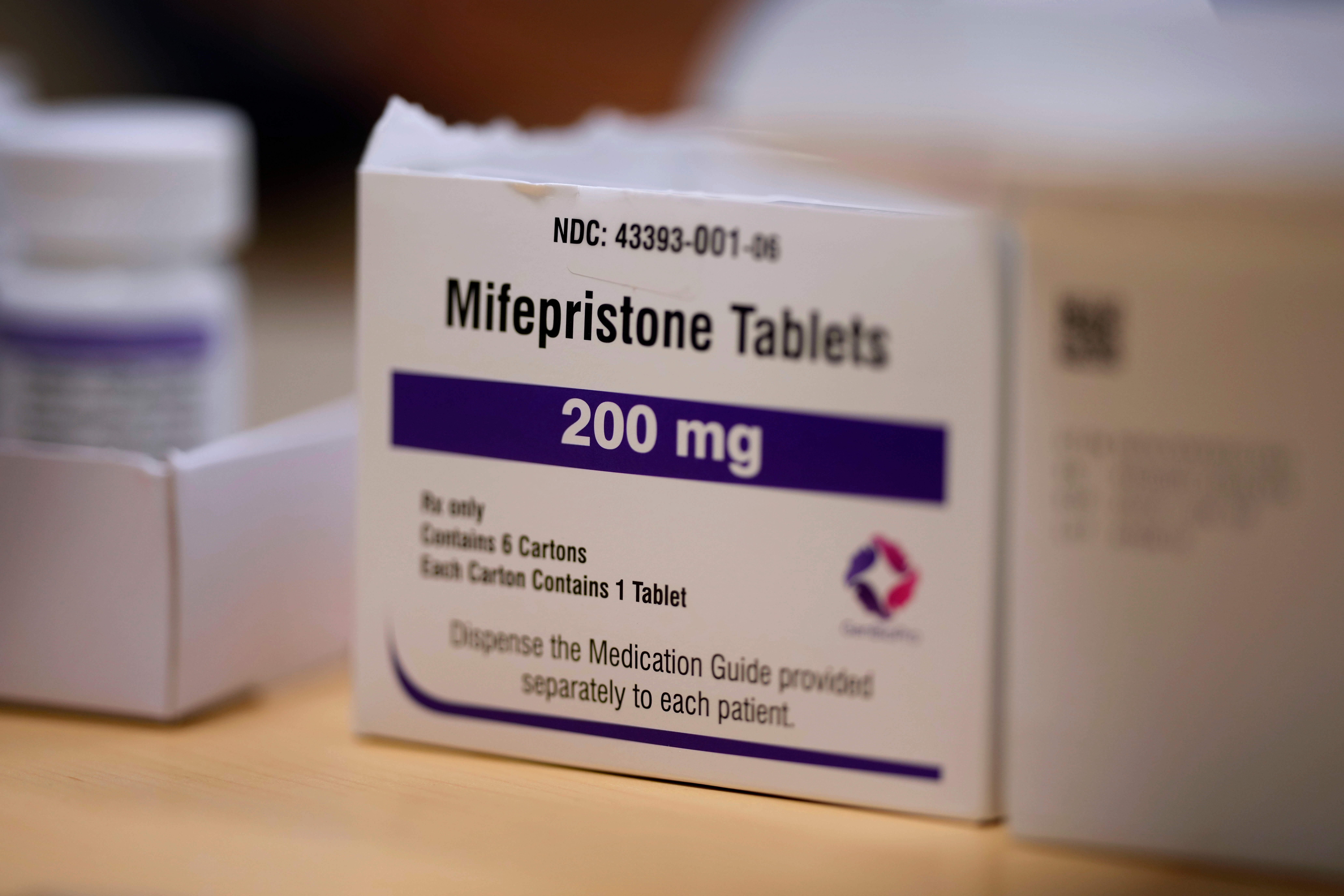
Health Court restricts abortion access across the US by blocking the mailing of mifepristone May 1, 20266:40 PM ET By The Associated Press Mifepristone tablets sit on a table at a Planned Parenthood clinic in Ames, Iowa, on July 18, 2024. Charlie Neibergall/AP hide caption toggle caption Charlie Neibergall/AP A federal appeals court has restricted access to one of the most common means of abortion in the U.S. by blocking the mailing of mifepristone. A panel of the New Orleans-based 5th U.S. Circuit Court of Appeals is requiring that the abortion pill be distributed only in-person at clinics. Since the Supreme Court's 2022 ruling that overturned Roe v. Wade and allowed enforcement of abortion bans, prescriptions by mail has become a major way that abortions are provided — including to states where bans are in place. The decision sets up a likely appeal to the Supreme Court. Sponsor Message Health Reporting in the States Abortion clinics are closing nationwide. Could urgent care help fill the gap? Health Over-the-counter medication abortion? These researchers say it would be safe A federal appeals court has restricted access to one of the most common means of abortion in the U.S. by blocking mailing of prescriptions of mifepristone. A panel of the New Orleans-based 5th U.S. Circuit Court of Appeals is requiring that the abortion pill be distributed only in person at clinics. "Every abortion facilitated by FDA's action cancels Louisiana's ban on medical abortions and undermines its policy that 'every unborn child is human being from the moment of conception and is, therefore, a legal person,'" the ruling states. Judges have long deferred to the Food and Drug Administration's judgments on the safety and appropriate regulation of drugs. FDA officials under President Donald Trump have repeatedly stated the agency is conducting a new review of mifepristone's safety, at the direction of the president. The judges noted in their ruling that FDA "could not say when that...